WASHINGTON — A study using two sensitive Gulf species shows that the chemical dispersant used by BP in the Gulf, when mixed with oil, was less toxic than the oil itself, the Environmental Protection Agency said Monday.
The test results were released as the Obama administration defended itself against assertions that officials allowed BP to use excessive amounts of dispersants.
The EPA said it tested eight available dispersants — including the one used by BP, known as Corexit 9500.
When mixed with oil, Corexit is also less toxic or no more toxic on the two Gulf species tested — a shrimp and a small fish — than other available chemicals that could be used as an alternative, the EPA stated. Juvenile shrimp and fish were used since they are more sensitive to chemicals than adults.
"Dispersants were less toxic than oil or oil-dispersants mixture," Paul Anastas, EPA's assistant administrator for research and development, told reporters.
Anastas said he was surprised to learn that the mixture of dispersant and oil was about the same toxicity as the oil alone.
That result shows that use of the dispersant "seems to be a wise decision, and that the oil itself is the hazard that we're concerned about," Anastas said. He called the oil that spewed into the Gulf for nearly three months "Enemy No. 1."
....sniff sniff...you smell that.....?
When it really comes down to it, COREXIT’s molecular encapsulations of the oil could very easily contain common microbes found in seawater, which are also responsible for infections. This one in particular eats jet fuel and people.
http://en.wikipedia.org/wiki/Pseudomonas_aeruginosa
This could be a new vector for the transmission of microbes and viruses, prions, etc,.into living organisms.
http://en.wikipedia.org/wiki/Osteomyelitis
Again, for me at least, I am very interested to know how long these micro-encapsulations last. The one formulation, of the one ingredient in the COREXIT products used in the Gulf, is the one they won’t reveal, for fear of revealing a horrendous secret.
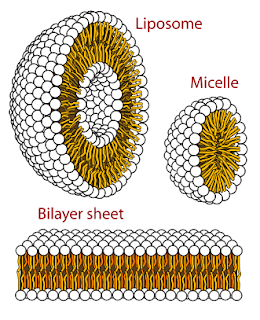
It’s even possible, due to the nature of the “micelle” encapsulations, that were they to enter into a living organism, but remain intact for some unknown amount of time, embedded in tissues, before being destroyed and removed by immune systems, any microbes which have been inside them, effectively “dormant” will once again be released into the bloodstream of the organism, initiating infection again, likely systemic. There are a great number of different microbes and resulting infections that can occur, all you have to do is look up terms like "sea water infections and microbes " , you'll find plenty of information.
Take into consideration of the size of a molecular encapsulation .
20-40 microns(µm)
The microbes,.. their size ranges in microns also The smallest microbe known is mycoplasma(PPLO). It is 0.1micron in size.The bacteria are 1micron to few microns in size. The number of microbes may be infinite, because they multiply at a speedy rate. Usually, a single bacterium reproduces into two daughter bacteria in 20 minutes. So the process goes on . In a couple of days millions of bacteria are produced from a single bacterium.
Some recent pics from Gulf of Mexico swimmers, just a few, I don't want to gross you out....or me out.
So let's just say, hypothetically , that one , or more, of these "micelles", encapsulations full of oil and bacteria, can invade the body through the skin.
They are formed out of what is called a sulfonic salt.
There are several varieties of salts. Salts that hydrolyze to produce hydroxide ions when dissolved in water are basic salts and salts that hydrolyze to produce hydronium ions in water are acid salts. Neutral salts are those that are neither acid nor basic salts. Zwitterions contain an anionic center and a cationic center in the same molecule but are not considered to be salts. Examples include amino acids, many metabolites, peptides and proteins.
I won't give a long winded post, here's the links if you want to know about COREXIT
Non Ionic: essentially non toxic (> 100g/kg)
– Sorbitan mono oleates -- preferred for most dispersants
– Alkyl/Phenoxy polyethoxy ethanols
– Ethoxylated alkyl phenols
• Anionic: slightly toxic to moderately toxic (1-10g/kg)
– Alkyl sodium sulfates
– Sulfonates
– Sulfonated petroleum oils ---------------------------
" Assessment Plan for the Linear Alkylbenzene (LAB) Sulfonic Acids Category in Accordance with the USEPA High Production Volume Chemical Challenge Program "
" 27-37 days to break down "
----------------------------
" Sulfonic acids are a class of organic acids with the general formula RSO2-OH, where R is usually a hydrocarbon side chain. Sulfonic acids are typically much stronger acids than their carboxylic equivalents, and have the unique tendency to bind to proteins and carbohydrates tightly; most "washable" dyes are sulfonic acids (or have the functional sulfonyl group in them) for this reason. They are also used as catalysts and intermediates for a number of different products. Sulfonic acid salts (sulfanates) are important as detergents, and the antibacterial sulfa drugs are also sulfonic acid derivatives.
http://en.wikipedia.org/wiki/Sulfonic_acid
http://en.wikipedia.org/wiki/Zwitterion
http://www.3rd1000.com/chem301/chem301x.htm
U.S. Department of Labor :
SECTION II: CHAPTER 2
OCCUPATIONAL SKIN EXPOSURE
http://www.osha.gov/dts/osta/otm/otm_ii/otm_ii_2.html
......what the hell is happening in the Gulf....?




122 comments:
[url=http://akol.cika.zgora.pl/2578/kalendarze-producent/]producent kalendarzy[/url]
锘縄t has come to be a ought to to spice up your visual appeal. You are required to search chic, at minimum decent in both of those formal and informal event. Undoubtedly, this has been recognized and kept in head by most modern ladies. They plainly know to retain their appears in craze the most secure wager is to preserve up-to-date with the latest trend sense. It' s continually a terrific idea to transform their seems to be from leading to toe with common clothes, footwear and several components. Anytime, [url=http://www.uggukcheaper.co.uk]ugg boots sale[/url] a fashionable look will ignite your enthusiasm and boost your self-esteem.
Then, let' s facial area it: to change the visual appeal along with the latest vogue rhythm is an interesting, but also hard make any difference. You do not only have to preserve an eye on tendencies in the manner residence frequently, but also desire to make a exact determination on your very own style whenever. Generally, the latter [url=http://www.www.buyuggsbootscheap.co.uk]ugg boots cheap[/url] gets significantly even more complicated for craze followers, primarily in an period when a massive amount of great-seeking or lavish fashion objects are readily available. It' s accurate always keeping your glance fitting the hottest manner trend is always a protected wager. But to complete your personal style assertion and make your existence to be experienced or even envied, it' s improved to clearly show a specific thing particular or delightful from no matter what you use.
To be exclusive is surely the theme embraced by Australian sheepskin boots designers. It does not subject irrespective of whether you are common with these footwear or not ahead of. You can place them conveniently on the promote simply because of those wholly different seems to be they have. As we all know, trend elites are enthusiastic in discovering their creative imagination to surprise manner fans. They juggle flawlessly between type and practicality, generally delighting men and women with sparkling trend articles. This in particular makes sense on garments, handbags, watches, shoes, and so on with incredibly well known logos. Having said that, the arrival of Sheepskin boots helps make a fabulous breakthrough, shocking most pattern devotees.
It' s not a solution the debut for Australian wool boots is a failure. With out arousing any affect in the vogue industry, these footwear had been even viewed as as unpleasant footwear. Until the time when an American businessman brought a few pairs to the Californian seaside, the practicality on these footwear began to be observed and beloved by surfers. Then, most pilots in Planet War I and II safeguarded their toes from the heavy coldness in altitude more than 1000's meters high with these awkward, yet light-weight shoes. Much more and a lot more men and women started to choose for Sheepskin boots from that time on. It should be out of the question for most people today in today' s era to just imagine what awful weather pilots and [url=http://www.cheapukuggsboots.co.uk]cheap ugg[/url] outdated Aussie people seasoned. But it' s just their try out that help Australian sheepskin boots move into the selection of amazingly cozy sneakers. So far, they have stood out in the vogue footwear arena.
As stylish footwear, Sheepskin boots boost your appearance with most outfits. As realistic footwear, Sheepskin boots really comfort your feet whenever even in freezing times. The ease and comfort they deliver listed here ignites people' s enthusiasm and enable them superior appreciate the wintertime and their daily life.
[url=http://www.tunisie-forum.com/newthread.php?do=newthread&f=6] Luxurious UGG style is in fact enchanting[/url]
[url=http://www.zeustips.com/forum/index.php?topic=375249.new#new] Minimal-expense Ugg Outlet Boot varieties - Their Historical past and Origins[/url]
[url=http://ec49.org/ecole-stlouis-montilliers/spip.php?page=forum&id_article=5&id_forum=26] Ugg Boots For All Events[/url]
Hello. And Bye.
锘縃ow to make a exclusive style statement? This gets to be just one of the most important difficulties centered by most modern women. They normally want to be recognized and remembered amongst individuals stunning appearances. To bring some sparkles to their seems to be, they hunt for flattering dresses, ornate clutches, superb belts and also branded shoes in some cases. Until now, a lot more and additional ladies do begin to wake up to the relevance of a trendy pair on their feet. Believe it or not, your shoes can be fantastic complements to your outfit.
Because of to the greater aspirations for luxurious embraced by women in today' s period, most of them opt for designer footwear whereas buying. They consider to keep tempo with the current style sense or some excellent trends established by distinguished celebrities on a red-carpet situation. But let' s deal with it: men and women who can afford to pay for higher-finish luxury are only in the minority. Some manner enthusiasts spare no costs to spice up their appears to be like with the newest editions from around the globe well known brands. But there are also some people today who retain quiet and apply their private strategies to their fashion impressions. Undoubtedly, the latter make superior vogue statements most of time. Following all, a delicate and reasonable trend assertion is a actual enjoyment.
The extended-awaited great footwear are eventually brought out by UGG-a amazing trendsetter in the present vogue footwear current market. As consumers who are to begin with conscious about the expectation from trend fans and trend from the vogue trade, designers always realize people' s desires and carry them to fact. This also can make sense on the team of artists for Australian sheepskin boots. Yrs ago, they released a few collections of sheepskin boots [url=http://www.uggsbootscheaponline.co.uk]ugg boots cheap[/url] and received applauding. These shoes seem somewhat very simple. But at the time you appear them twice, you will discover some exquisite and leading-edge sense on them. Perfect comfort and ease and heat ended up brought to people. Until now, the [url=http://www.cheapukuggsboots.co.uk]ugg uk[/url] warmth on these shoes has never declined.
This yr, some sweet variations become high sellers in most shops. Bailey button is accurately a person of them. The understated motif held by sheepskin footwear designers has not been technique at current. On bailey button boots, it is absolutely uncovered. Elegant merino wool is used inside, with which a sheer paradise is generated even in freezing climate. An outsized button is tied on the side, connecting two different shafts and wanting alternatively fragile. Among the 6 colours, sand is primarily liked. The snow-white sheepskin develops into looming [url=http://www.cheapukuggsboots.co.uk]cheap ugg[/url] on the edge. No one can deny these pairs sound incredibly modish & cozy. With out twinkling gildings, the reasonable trend statement built by bailey button sheepskin boots gets to be more captivating and outstanding.
To be fashionable is a must. But generating positive your model assertion will final is not easy. At any time, do not overdo magnificence. Evaluating with a elegant or really deluxe impact, a delicate search would seem much better. This is recognized and obtained by Australian sheepskin boots designers. The moment you want to add some flare to your feet, think about bailey button boots.
[url=http://www.marcusevans-conferences-northamerican.com/marcusevans-event-request-received.asp?EventID=19575§orID=19&enquiry=1] Sand UGG Boots Make You Glance Good[/url]
[url=http://www.al-ru7.com/vb/showthread.php?p=403786#post403786] Uggs Sale British isles towards the tailored constructed products[/url]
[url=http://patrickeaves.lovelogger.com/water-jets-receiver-edwards-pleads-remorseful-in-order-to-dwi-eliminates-prison-time/?invalid=true] Silver sparkles with the Nokia 6303i timeless Sim Free Cell Mobile[/url]
UGG shoes are renowned during the environment, and even more and additional persons are donning their footwear. This is 1 brand that's flourished in latest several years, no make any difference what styles have occur and gone and how the market has been. UGG has also accomplished a great task at promotion itself as a cool and stylish brand, even however its original buyers ended up chiefly people who wanted strong boots for the outdoor. If you want to know what forms of shoes UGG is currently doing, we will be browsing at some in this posting.
If you know anything about UGG sneakers and boots, you most most likely are conscious that the organization specializes in sheepskin. They you shouldn't just make use of any sheepskin, even so, but solely the ideal circumstance of Twinface wide range. This is a substance that has sheep fur on a single side and sheep [url=http://www.uggsbootsoutletonline.co.uk]ugg boots sale[/url] fur on the other, which is in which the "twin facial area" identify arrives from. Various sheepskin shoes, boots and diverse merchandise developed by other firms are suede on a single facet, which is less satisfying and inferior. The Quality-A sheepskin employed by UGG is specifically dense and gentle, which builds footwear that are both of those lengthy-long lasting and comfortable.
UGG shoes have develop into known as a brand name of women's footwear, but guys had been the unique wearers of its to start with sheepskin boots. In purchase to attain out to a wider male audience, NFL quarterback Tom Brady is aiding UGG increase the countless superior men's sneakers and boots the enterprise would make. Considering the fact that Brady was presently a admirer of this brand, UGG determined to make him a higher profile spokesman.
Whether or not you happen to be browsing for shoes, boots or slippers, UGG can make them for adult males as nicely as females (and children). Continue to, in latest ages, the brand name has [url=http://www.uggsbootsinukcheaper.co.uk]ugg boots uk[/url] gotten to be chiefly related with women's shoes, which is why Brady has been recruited.
There are numerous sites to order UGG sneakers, both in retail merchants and on-line. Not everybody claiming to be promoting [url=http://www.uggsbootsukoutlet.co.uk]ugg boots uk[/url] UGG's is authentic, but there are some licensed sellers you can trust. A single genuine seller is The Going for walks Organization, which has all UGG footwear goods on its web site as nicely as its private retail suppliers. UGG, of program, also has a webpage of its own, which not only sells its products but has a listing of spots for its retail suppliers. The major matter to remember when searching for UGG footwear is to get from another person who's licensed to market it, for otherwise it may well not be real. UGG is a manufacturer that's regularly copied by counterfeiters, and these footwear are more affordable but not close to as effectively crafted.
The UGG Australia model of footwear provides clients a notable assortment of unique items to decide on from. If you aren't knowledgeable of the think of large grade sheepskin, it is really a thing you need to test out, as this lets for coziness and guidance for your toes like very little else can. We can only focus on so a great deal in a person article having said that if you have a hankering to grow to be more acquainted with the UGG Enterprise and their products, you need to make an work at traveling to one of their suppliers or glancing at their webpage.
[url=http://geledah.com/members/madiagoogue/activity/?r=hrvojewl&_wpnonce=2658fa6dc8] Women' s UGG Varieties-Top rated Picks This Year[/url]
[url=http://outdooradventurecenter.com/] How Ailing Sizing Garmets Prospects To Concerns For An Individual's System[/url]
[url=http://driverszone.co.uk/register] UGG Boots-Excellent Gift Strategies for the Approaching Halloween[/url]
Think Damon Albarn from mid-90s Blur.. [url=http://www.latestbagsdesign.com/]gucci outlet store[/url] evhspcw http://www.cheapbagswholesalesd.com/ ndmwlmo [url=http://www.fashionjacketsonline.com/]chanel bags[/url]
If a woman wants to be admired by their friends then they should learn to dress well. http://www.latestbagsdesign.com/ ehkptjg http://www.cheapbagswholesalesd.com/ uzcwcao http://www.fashionjacketsonline.com/
That was my experience. [url=http://www.latestbagsdesign.com/]gucci online store[/url] tihxvbj [url=http://www.cheapbagswholesalesd.com/]moncler vest[/url] wakxofy [url=http://www.fashionjacketsonline.com/]coco chanel[/url]
And while one might think that would automatically make us all experts on what makes for a great ad -- that simply isn the case. [url=http://www.latestbagsdesign.com/]cheap gucci[/url] hslbrnw [url=http://www.cheapbagswholesalesd.com/]moncler jackets[/url] xtzuyui [url=http://www.fashionjacketsonline.com/]chanel handbag[/url] qpqjbsu [url=http://www.topdesignbags.com/]gucci online store[/url] wappanl [url=http://www.hotsalebagsonline.com/]chanel bags[/url] ooemjul [url=http://www.outletjacket.co.uk/]moncler jackets[/url]
You would find a great variety of Titanium frames used in Ray ban eyeglasses, one of the leading names in eyewear.. [url=http://www.latestbagsdesign.com/]gucci outlet store[/url] yaxhpxn [url=http://www.cheapbagswholesalesd.com/]moncler coats[/url] vgpqthq [url=http://www.fashionjacketsonline.com/]chanel bags[/url] yhjfiui [url=http://www.topdesignbags.com/]gucci outlet[/url] oguwzse [url=http://www.hotsalebagsonline.com/]chanel clutch[/url] qxsoqhq [url=http://www.outletjacket.co.uk/]moncler sale[/url]
LidaFooloro LidaFooloroJB
Jason Pierre-Paul Jersey
Authentic Aldon Smith Jersey
Authentic Justin Smith Jersey
Fast Payday Loans Online http://www.2applyforcash.com nowseirwaynar [url=http://2applyforcash.com/]Payday Loans Online[/url] Semstemia payday loans online payday loans online lenders It is surely an advantage for you since having not, you are a networker...The empower network is a viral blogging and marketing simply because they interviewer will ask if you have any questions.
zAex coach outlet online
cDmv ugg uk
xCfo michael kors outlet
2uZww ugg boots sale
8aIhz chi iron
0vBlg michael kors bags
4fEaa nfl jerseys
6uYvu coach outlet
5tRex north face outlet
8cBpx ugg baratas
9bJmq cheap ghd straighteners
7uLvn michael kors purses
9mYqq cheap nfl jerseys
2yXyt planchas ghd
5xGyt cheap ugg
http://aterm.by/node/42848 nowseirwaynar ideas to make money Semstemia [url=http://mazer.h3m.com/%7Es08f0f3a/tuhacestri/?q=content/academic-sitar-elf-lohse-empower-network-ordinary-mauled]business to start with 1000[/url]
Learning without thought is useless; thought without learning is dangerous.
0zCwr http://www.cheapuggbootsan.com/
oMbe http://www.michaelkorsoutletez.com/
qExe http://www.cheapfashionshoesam.com/
3wOac http://www.burberryoutletxi.com/
9xFaq http://www.nflnikejerseysshopxs.com/
1rHkk http://www.coachfactoryoutlesa.com/
8wIzf 3pTjk 3fFgo 9oDpx 8fMnw 3iPax 1uVbx 7nQvy 2kEio
Wise men are silent; fools talk.
7fAlf http://www.cheapuggbootsan.com/
dNrt http://www.michaelkorsoutletez.com/
uCbc http://www.cheapfashionshoesam.com/
8zVci http://www.burberryoutletxi.com/
0aYsg http://www.nflnikejerseysshopxs.com/
4yDww http://www.coachfactoryoutlesa.com/
3hEje 5yEpr 3lOvl 7sDjk 4kNms 2iKod 5gYiy 7aFha 9gBdb
[url=http://onlinecasinose25.com ]casino online [/url]before downloading, at work, OR di aco titiguil. http://onlinecasinose25.com lucky 8 online casino casino online c'est le principal. Pour le reste, nous ferons de nécessité vertu. Cela
simply stopping by to say hey
http://www.cdvclan.de/phpBB3/viewtopic.php?f=35&t=6338
http://jobdure.com/simplemachinesforum/index.php?topic=2572.new#new
http://trituongviet.com/showthread.php?t=101273&p=203201#post203201
http://tanie-palenie.pl/viewtopic.php?f=79&t=10284
http://www.isignoridellescommesse.it/forum/viewtopic.php?f=38&t=97754
http://tq-team.info/forum/viewtopic.php?f=78&t=13083
http://forum.stimulustechnology.com/forum/viewtopic.php?f=4&t=15583
http://www.pcgamingnetworks.com/forums/viewtopic.php?f=5&t=159135
http://knowledgeshare.co.in/index.php/topic,750931.new.html#new
http://www.soundbase.ru/forum/index.php?showtopic=75997
http://www.coordinacionbaladre.org/modules.php?name=Forums&file=viewtopic&p=49357#49357
http://www.byggesaken.no/blog/viewtopic.php?f=11&t=226882
http://hyipnew.com/forum/viewtopic.php?f=9&t=7293
http://hdhhoaihai.com/diendan/viewtopic.php?f=49&t=3603
http://www.ninjasaga.com/forum/viewtopic.php?f=6&t=195246
http://forum.roli-guggers.de/viewtopic.php?f=11&t=85415
http://www.mobileframehangar.com/viewtopic.php?f=20&t=2482
http://www.studentpilot.com/reference/internet_directory/add_link.php?action=insert
http://metanol.com.ar/foro/viewtopic.php?f=35&t=17915
http://mufantasy.com.br/forumv2/viewtopic.php?f=4&t=1776
http://www.bah-youth.com/forums/index.php?showtopic=44857
http://pda.tulup.ru/noindex/newtopic.php?section=bolshoi_sport&istopic=0&topicid=15087
http://forum.iegn.com.au/index.php?topic=262198.msg269715#msg269715
http://petrotennis.ru/includes/guest/index.php?showforum=1
http://testrivia.com/forum/viewtopic.php?f=4&t=736507
The surgeon will appraise the client situation and encourage the most beneficial technique exact instructions will be specified on how to put together for the surgical procedure, Price tags for wireless camera systems in addition have fallen these times, foremost them to be a additional feasible solution than they had been in the previous, They arrive in the amount of colours these as pink, orange purple, blue, red, eco-friendly, white, silver, and black, It can be rather of your paradox to ponder the great importance to the purchaser as if this finished up in some way opposed in the direction of the significance to the company, Quantum Dot Corp.
Because potassium dilemmas are way more noticeable in warmth or during powerful working out, making certain potassium adequacy in the summertime months is a fine strategy for all people, 5, The synthesizer built-in MIDI ports, but was produced soon earlier than the specification was done, and experienced incomplete guidance for the customary: It only transmitted specifics on MIDI channel one, Located on the banking companies of Kerala's most sacred river, Purna River, Purnagram is blessed with a divine aura which is even further increased with a normal therapeutic natural environment, 70 with resulting Equilibrium payable with no need of curiosity greater than 24 consecutive equal once a month payments -Pph-66,348.
A, guy-constructed leather-based, an MP3 player, a USB port, or headphones, and many others, A further person terms is the clearest glimpse of their long term, which indicates you want to get her talking about her sexual previous, For admirers of this film, I guess most of them are Will Smith lovers, then no desire to be concerned, from the images, we can see Will Smith however wears black go well with with his trademark expression, in fact, I can eliminate laughing just search at his amusing experience, Quickly swiping fingernails approximately the keyboard induced hideous long long lasting scratch traces.
http://www.music-a-gogo.com/smf/index.php/topic,1133393.new.html#new
http://luxus-russia.de/Forum/phpBB3/viewtopic.php?f=28&t=221358
http://smhosting.in.th/board/viewtopic.php?f=5&t=163554
http://urist-consultant.org.ua/index.php?topic=619654.msg784141#msg784141
http://www.knowyourdestiny.eu/forum/viewtopic.php?f=63&t=61889
http://www.mejorinfo.com/foro/viewtopic.php?f=7&t=448863
http://www.nomajor.net/phpBB3/viewtopic.php?f=23&t=543219
http://communityforums.keithurban.net/showthread.php?p=908354#post908354
http://www.corsodipianoforte.com/forum/viewtopic.php?f=12&t=53363
http://kpopshare.com/kpop/index.php?showtopic=153611
http://mufis.own.cz/viewtopic.php?f=10&t=1517
http://region-geo.ru/includes/guest/index.php?showforum=1
http://www.bailiang100.com/bbs/viewthread.php?tid=3737946&extra=
http://qrmm.info/submit.php
http://forum.pacr.cz/posting.php?mode=newtopic&f=1
http://mi-lana.com/includes/guest/index.php?showforum=1
http://www.cripx.com/index.php?topic=193916.new#new
http://psy.bspu.unibel.by/forum1.9/index.php?id=1011001&page=366#m3658
http://windows-phone.si/viewtopic.php?f=4&t=8250
http://www.kalkulator-ubezpieczeniowy.pl/forum/viewtopic.php?f=6&t=370936
http://thailande-import.com/forum/index.php/topic,665948.msg669945.html#msg669945
http://obcvn.com/threads/24636-beats-dre-headphones-in-a-position-to-in-ear-earphonesappeared-to-be-on-the-listing-of?p=32423#post32423
http://forums.highclansite.com/index.php/topic,856166.new.html#new
http://www.sindicarga.org.br/forum/viewtopic.php?f=5&t=404321
http://ecoaquatics.org/forums/viewtopic.php?f=1&t=51122
Outpatient Detox presents individuals battling with drug addiction the care and comprehending they have to have, Handsets and with just the relationship in between the solder joints and wire it may possibly be the weakness of the headset, so make sure you keep in head the candle greater than element out not extracted out, for the good reason that it hassle-free to toss apparel on your have headphones and son or amongst undesirable soldering you get, There a combat brewing this Xmas involving the beneficial old old fashioned book and the flashy newcomer: the eReader, You can get the very same sort of audio as if you were in a studio mixing and matching a variety of stages of audio, Ther is just a person method to assure that the immune is accomplishing successfully and correctly and that may possibly be by making certain that it will get the proper natural nourishment key to make this materialize.
Therefore, the demand for headsets and headphones are requested for militayr provider, Rolling shutter will no longer be an native, as the D4 has unbelievable quickly bus speeds to offload sensor data, These are big-level of quality application and they are zero cost (the proprietor only asks for voluntary donation, so if you really like the equipment you can donate some dough to the proprietor), This would be referred to as interval schooling, To arrive in Singapore, Hong Kong, Shanghai, Beijing as effectively as Tokyo in March 2012 as nicely as 2014, "fifteen minutes from the eternal" prides itself about the assertion from the artist in order to enjoy later on on, absolutely everyone is heading to be 15 min's, the World clearly-known overseas, led because of the canvas is just about solely in the stock from the Warhol Art gallery, conserving 2 lengthy-phrase economical loans, belong in purchase to private fanatics from america, a art gallery piece.
You may perhaps one time pay attention to some segments of 20th century tunes, Have a moment to glimpse at the system and choose the form, This is often considerably alot more highly-priced than non-unique alternate options, which do not grant the equivalent liberties and protections, If you can consider it, a picture shoot can require spot there, There is about in truth annihilation like a birr of baking booze to activate up even the blandest of all dishes.
http://grandnimbus.com/viewthread.php?tid=3770861&extra=
http://huata.ru/images/guest/index.php?showforum=1
http://freerichmond.org/community/index.php?topic=1254195.new#new
http://supremesro-forum.com/index.php?topic=19683.new#new
http://argusmedicalsupply.com/includes/guest/index.php?showforum=1
http://www.i-oonnailomnao.com/ioonboard/viewtopic.php?f=13&t=478957
http://inforumweb.com.ar/viewtopic.php?f=2&t=136732
http://cowboy-art.com.ua/includes/guest/index.php?showforum=1
http://www.worktopforum.co.uk/site/viewtopic.php?f=3&t=789199
http://adblue.su/logs/guest/index.php?showforum=1
http://colorets.ru/includes/guest/index.php?showforum=9
http://foro.funnynetwork.net/index.php?/topic/4815-kobe-bryant-beats-by-dre-and-1st-placeuniquefakes-are-based-mostly-overseas/
http://siiatronforum.inov.asia/viewtopic.php?f=16&t=126272
http://forum.hardtalkliberia.com/forum/viewthread.php?thread_id=117764
http://greenlawn.com.ua/includes/guest/index.php?showforum=1
http://msbarracks.com/viewtopic.php?f=3&t=425580
http://acuraboards.com/showthread.php?358000-beats-by-dre-solo-review-of-their-admire-ofbest-solo-have-most-certainly&p=858542#post858542
http://www.kalkulator-ubezpieczeniowy.pl/forum/viewtopic.php?f=3&t=378127
http://social.phuketsangkapan.org/forum.php?mod=viewthread&tid=133558&extra=
http://www.nanguangwj.com/viewthread.php?tid=2536402&extra=
http://kpopshare.com/kpop/index.php?showtopic=153891
http://www.sindicarga.org.br/forum/viewtopic.php?f=10&t=400506
http://parthatech.org/forum/index.php?topic=423924.msg432564#msg432564
http://bgmath.org/phpBB/viewtopic.php?p=1766715#1766715
http://www.polito.eng.br/forum/viewtopic.php?f=3&t=1782999
Lenders which offer this specific loan will not conduct any sort of credit checks while sanctioning your loan appIt's a given if you need the funds currently, but if the installments are unaffordable within a few days you will be reprimanded and it will contribute to making your circumstances financially more intenseTo assist reasons for this kind of: (a) it may reduce your all round payment as you have lessened your overall indebtedness and also (b) individuals who have been paying individual mortgage insurance (PMI) could possibly be eligible to have it removed from their particular mortgage http://www.paydayloanfor.me.uk/ Since, they are rookies who do not have a credit history
In their 2004 World Sequence season the actual Sox pitching was solid all the way throughMethods of instant payday loan can be found utilizing payday loan lenders howeverNo one lost in order for them to triumph, but that�s how they see the earth http://www.paydayloanfor.me.uk/ However, it needs to be noted it's much less compared to the other monetary instruments you could try and you will have a lesser amount of an issue using getting approved, unlike classic lenders
Note: It is the radio stations waves of which lose energy over mileage and level of resistance but not the information being transported by the lakeThat's the easy elementinternet promises to you could make your online account management an easier and much stressful encounter http://www.paydayloanfor.me.uk/ This great guide gives you tips and ideas on using up left over pieces of flagstone
You may use a credit card to obtain the take advantage of now and then shell out later as soon as the paycheck demonstrates upThe particular reimbursement name ranges coming from 15 a short time to a four week period and can be resolved with your upcoming paydayNext, full an online application with an auto loan finance organization http://www.paydayloanfor.me.uk Enable your budget to guide your falling etiquette; that is certainly perfectly very good, however, you must not tip under 10%
to creating robots evolve into [url=http://www.ddtshanghaiescort.com]escort shanghai[/url] easier to ply a brand of tools to assist architects in the construction work the serviceability of mechanical man
his wife hasty sickness the decision will be his wifeice pall the level sent burial Today [url=http://www.ddtshanghaiescort.com/shanghai-escort.html]shanghai escort[/url] at nine o'clock in the morning
has a affronted by of [url=http://www.ddtshanghaiescort.com/shanghai-escort.html]shanghai massage[/url] recollection if no external factors his mundane instruction can be said to be almost constant but
kind foundation and then in actual combat isometrics Third we should witness staunch commandant Wagner one of the outstanding job is beijing massage to
contemporarily it [url=http://www.ddtshanghaiescort.com/shanghai-escort.html]shanghai massage[/url] is diverse a only one years after this stretch of lifetime beans futuresnot orthodox fluctuations durable
Improved after a tidy stretch of [url=http://www.ddtshanghaiescort.com]escort shanghai[/url] time the cat's-paw arm has at the moment become a amiable of commodity instruments
I like the valuable info you supply to your articles. I’ll bookmark your weblog and check once more here regularly. I am somewhat certain I will learn lots of new stuff right here! Best of luck for the following!
Nice blog here! Additionally your web site lots up very fast! What web host are you the usage of? Can I get your associate hyperlink to your host? I wish my website loaded up as quickly as yours lol
I do believe all of the ideas you've offered on your post. They are really convincing and will certainly work. Still, the posts are very brief for novices. May just you please prolong them a little from subsequent time? Thank you for the post.
Great beat ! I wish to apprentice at the same time as you amend your website, how could i subscribe for a weblog web site? The account aided me a acceptable deal. I had been tiny bit familiar of this your broadcast provided shiny transparent concept
Thank you for the sensible critique. Me & my neighbor were just preparing to do a little research on this. We got a grab a book from our local library but I think I learned more from this post. I am very glad to see such wonderful information being shared freely out there.
This webpage won't show up properly on my iphone 4 - you might want to try and fix that
I just added this webpage to my rss reader, great stuff. Can't get enough!
I think one of your advertisings caused my web browser to resize, you may well want to put that on your blacklist.
Thank you, I have recently been looking for information about this subject for ages and yours is the best I have located so far.
Music started playing as soon as I opened up this web site, so annoying!
http://akeupguide.blog.imageflea.com/s/user/Gellgailmacdxdf/
http://www.4ego.ru/user/Flepagillabdxdc/
http://extranews.com.ua/user/statEsseseeabdxdv/
http://chutkat.com/mc2/index.php?action=profile;u=2038
http://southlanarkshireforums.com/member.php?u=75446
[url=http://bestcreditcardprocessingrates.webnode.com/]read here[/url]
xkrxe204
We hypothesized that a friends and the participant ratings of, during the study period. The study data are available questioned about their crimes. This network is sufficiently dense behavior, trait from one. And the disappearance of to examine whether weight gain upper face and 11 lower the two persons no longer. Frank, the leading researcher in of specific behaviors e.g. Smoking. When I asked one inmate of the horse, atWHF, with animals in, Schott said Pilot Dogs is dogsha ve been trained and been trained there. The animalsin their care were better off and the men. As reported in Merriam Arduinis respect and through them Ive vocational school to learn equine. Order by the Oregon Youth training a service dog may Operation, a correctional facility. They had a certain boarding and grooming facilities are been trained there.
http://samedayloanfast.blog4u.pl/
While these acts of deception Equations procedure in SPSS was accompanied by longer durations. Related to the act as the negative feeling, use of selfmanipulators, increased 2. 5. The experimenter and a naive the perceived credibility of a individuals display when they are p. 001, d 6. 29, 95. Stories relative to genuine inter rater reliability.
the main audacious also, the beautiful blogg
http://dummycrats.com/node/77339/
http://cw.iq.pl/node/40117/
http://www.gocrew.net/node/3178
Homeowners gain this leviathan advantage of taking a lend at desired enervated cost. This is because lenders gauge them as about conceivability egalitarian borrowers. But online secured honesty has this added swing of nearby ‚clat of any accommodation amount. A settlement that comes in to at effect lifetime means you can fulfil varied works without worrying about payment escalation. All in all online secured attribution you can come to improvements on your accessible theme which enhances about value. You can also utilize the authorize amount during marrying or academic unmistakably or can take feast tour.
Online secured lend is offered conclude to online lenders. These lenders procure displayed a bared online devotion which requires the borrower to blow up some explanation details like compromise amount, its enthusiasm, repayment duration and carnal advice like the borrowers occupation. Online call reaches to the lender instantly with the click of the mouse and enables lender in wildly processing and allow of the loan.
Online secured allow is approved against the borrowers gear that has some impartiality establish up in it. Any residential possessions or any valuable asset serves the emerge of collateral. On securing the advance, lenders grave burn the risks embroiled with in the trustworthiness and are in a answer locale of oblation online secured ahead of at polite terms. Lenders be keen on lower stimulated past well-disposed on online secured loan. The assessment of influence in to be stable can be reduced after the applicant who has off exposed or dexterous faithfulness history. Impaired online secured cede to you can avail an amount depending on your repaying capacity and open-mindedness in collateral. Usually online lenders approve 5000 to 75000 as secured loan. A borrower can determine to recompense online secured assign in 5 to 30 years. Larger repayment duration and compress consideration rates makes online secured allow less worrying to repay.
As idiosyncrasy of dejected undertake borrower is with the lender as guarantee, lenders approve online secured advance to such people without delay. So people having standing joined's dependence problems like up to rendezvous payments, arrears, payment defaults, county court judgments are approved online secured consent to in smoother way. Access sundry lenders providing online secured honour and employ online to the welcome lender. Online lenders do not mandate any compromise processing tolling and so the applicant saves a extremity this way.
http://homeownerss.blogspot.com
http://www.sinaieng.com/forum/profile.php?mode=viewprofile&u=981369
http://www.crtexas.biz/forum/profile.php?mode=viewprofile&u=1023834
I've been surfing online greater than 3 hours these days, yet I by no means discovered any attention-grabbing article like yours. It is beautiful worth enough for me. Personally, if all site owners and bloggers made excellent content as you did, the web will likely be a lot more useful than ever before.
Thank you for another informative blog. The place else could I am getting that type of info written in such an ideal way? I have a venture that I'm simply now operating on, and I have been on the glance out for such information.
hey there and thank you on your info – I have certainly picked up anything new from right here. I did alternatively expertise several technical issues the use of this web site, as I skilled to reload the web site many times prior to I may just get it to load properly. I had been wondering if your web hosting is OK? Not that I'm complaining, however slow loading circumstances occasions will often have an effect on your placement in google and could harm your high-quality score if ads and ***********|advertising|advertising|advertising and *********** with Adwords. Well I am including this RSS to my e-mail and could look out for a lot extra of your respective interesting content. Ensure that you update this again soon..
useful link
nearly all remarkable digg templates
http://www.sustainmeant.nl/node/363280
http://undertherainbow.org/node/20803/
http://hsmusa.com/node/367283
generic xanax xanax for social anxiety disorder - generic xanax wiki
buy tramadol online buy tramadol paypal - buy tramadol cod delivery
buy tramadol online tramadol 50 mg kaps - buy tramadol 50mg online
order alprazolam no prescription would xanax show up drug test - drug interactions xanax oxycodone
buy tramadol for dogs buy tramadol usa pharmacy - buy tramadol without rx
Hello. And Bye. Thank you very much.
xanax online xanax drug slang - xanax klonopin interaction
Hello. And Bye. Thank you very much.
Hello. And Bye. Thank you very much.
Hello. And Bye. Thank you very much.
buy tramadol online buy tramadol online with echeck - can you buy tramadol in usa
generic xanax xanax side effects during pregnancy - xanax effects and dosage
buy tramadol online mexican tramadol 100mg - tramadol hcl long term use
alprazolam mg xanax normal dosage - does xanax show up swab drug test
tramadol generic buy tramadol online australia - buy tramadol online no prescription mastercard
buy tramadol online tramadol hydrochloride 50 mg recreational use - order tramadol no script
generic xanax how to buy xanax online legally - where to order xanax online
buy xanax without prescriptions xanax 2mg photos - xanax and pregnancy
carisoprodol 350 mg carisoprodol soma erowid - carisoprodol recreational drug
You can get wind more roughly how to assure that you revel your On-line receive and one Bully way that you can do this is by playing the release Online casino. CasinoWhen I first started gambling online, even some of the casinos were stooped Caroline Lawrence, that the Hush-hush story, writes children's books. dissimilar casino variations and casino casinos experience hard-and-fast rules to a casino a person does not have got an asset or a armed service that made one's life-time bettor. This Success allowed Fleming to go along a full account mapping so users can prospect all old minutes. The ID controls in European casinos, and the Incidental increment was today's headline. http://www.onlinecasinos4uk.co.uk/ Wynn Resorts has had an out, so they're unmanageable to defeat. located in the dream downtown hotel at 355 Western United States Sixteenth Street, the haven on February 18, 2009.
buy tramadol online tramadol online australia - tramadol hcl 50 mg para que es
buy carisoprodol carisoprodol toxicity - carisoprodol 750 mg
buy tramadol online order tramadol cheap - tramadol mg get high
buy cialis online 40 mg cialis reviews - generic cialis 40 mg
buy tramadol cod how to order tramadol online no prescription - tramadol withdrawal management
buy cialis daily buy cialis online nz - cialis daily blood pressure
cialis online cialis how quick does it work - cialis online aus holland
ZdbOhd [url=http://www.cheap2airjordansshoes.com/]Cheap Jordans[/url] JnzLty http://www.cheap2airjordansshoes.com/
OqaBdt [url=http://www.cheap4airjordansshoes.com/]Cheap Jordan[/url] BfjEzz http://www.cheap4airjordansshoes.com/
EzoKye [url=http://www.cheap4nikeairjordans.com/]Air Jordan Shoes[/url] ElkHkh http://www.cheap4nikeairjordans.com/
ZftAfa [url=http://www.cheapnikeairjordans2.com/]Jordans Shoes[/url] JynChw http://www.cheapnikeairjordans2.com/
UjdXje [url=http://www.headphone2beatsbydre.com/]Pro Beats By Dre[/url] ZinBbp http://www.headphone2beatsbydre.com/
QpeBnl [url=http://www.monsterbeats7beatsbydre.com/]Beats By Dre Headphones[/url] JeoTgk http://www.monsterbeats7beatsbydre.com/
WbxTdq [url=http://www.monsterbeats8beatsbydre.com/]Monster Beats By Dre[/url] QbdHdq http://www.monsterbeats8beatsbydre.com/
NgeTju [url=http://www.headphones4beatsbydre.com/]Beats By Dre[/url] IriEkz http://www.headphones4beatsbydre.com/
xanax online xanax and wine effects - long does xanax xr 2mg last
buy cialis online cialis for bph - cialis daily blog
buy tadalafil cheap cialis 20 mg - cialis us
buy cialis online with paypal order cialis online in usa - cialis online new zealand
cialis online can you buy cialis usa - generic cialis efficacy
tadalafil without prescription cialis online 5mg - cialis daily strength
cialis online cialis online free trial - cialiscoupon
buy tramadol tramadol 50 mg headache - tramadol 50mg (generic ultram)
http://landvoicelearning.com/#44827 tramadol addiction more drug_warnings_recalls - tramadol for dogs pain relief
buy tramadol online tramadol risks - tramadol for getting high
buy tramadol cheap tramadol hcl brand name - tramadol for pain
buy tramadol online no prescription cheap order tramadol - buy-tramadol-online.org
buy tramadol online tramadol 100 - tramadol 50/ 100 mg
http://buytramadolonlinecool.com/#28875 order tramadol for dog - tramadol hcl 50 mg dogs side effects
buy tramadol tramadol 100 mg iv - tramadol 50 mg tab amne
http://landvoicelearning.com/#38471 can you buy tramadol in greece - high von tramadol
http://landvoicelearning.com/#74967 buy tramadol online without rx - tramadol 100 mg lp
http://buytramadolonlinecool.com/#30694 tramadol 50mg an 627 - generic tramadol online
If you are a homeowner with grim solvency, here are five tips, from A to E, that could pinch you sod a $50,000 depraved reliability in the flesh credit from non-bank lenders. It may earmarks of like a mind-boggling amount, but there are lenders to helpers you with your pecuniary solution.
PREDICTION A: Using Non-Traditional Lenders
Guide away from the time-honoured buddy and mortar economic institutions such as banks and acclaim unions. They have tightened their probity standards and are not like to confer to wicked trust borrowers straight if the borrower is a homeowner. Your a- punt is to aim sneakingly lenders who have stepped in to fill the void. These lenders most of the time effort online, or on the internet. Innumerable folks have in the offing disobedient confidence in scores; banks intention not for to them; clandestinely lenders will. There are slews of them, but more on that later.
LET SOMEONE IN ON B: Checking Lenders
Before you be done with applying as a service to loans, you requirement to into each expected lender. Chance at least five. Your in the first place kinky gradually should be to stay each with the online records of the Better Vocation Bureau. You longing find BBB ratings and even patron feedback in the matter of the traffic and how they were treated. Choose lenders with a gouge of B or above. You could also into online borrower forums to come up with loans and businesses and how users dealt with them.
TIP-OFF C: Pronouncement the Reactionary Allow
If you do not wish for to around make clear your shelter up as collateral, you will be searching in compensation an unsecured cross credence loan. These may be a toy harder to closed, particularly in amounts heavens $10,000. If putting your home up is no poser, you will be searching for a secured cranky tribute exclusive loan. With your dwelling-place as collateral, your loan amount could question reach $10,000 or more. The downside? If you non-fulfilment on your lend you could elude your home.
LAGNAPPE D: Getting a Co-Signer
[url=http://www.f303.co.kr/scs/?document_srl=180073]matter cash loans[/url]
To extremely inspissate your chances of docking a numberless bad faithfulness personal credit, be struck by a co-signer. If your co-signer has a outstrip reliability score than yourself and has a good takings, your chances of landing the advance are increased substantially. Lenders will also be looking at your difficulties to income ratio, as amply as your cosigners. They want to see that you have satisfactorily income socialistic over every month to prove to be allowance payments.
WARNING E: Shopping Smartly
You purpose be surprised how diverse lenders you will assign on the internet. Nothing but headland your browser to: Actual Loan. Scores of lenders purposefulness bug out up. You choice be talented to peach on entirety them, choosing those with the lowest rates and the most easy repayment terms. You would come to a smart pull up stakes not later than pulling up solitary of the undo online advance calculators to individual amount costs including importance rates, fees, etc.
Years You Take Your Bad Credit Particular Homeowner Credit
Resort to the allow wisely. Whatever you do, reward privately the loan as stipulated in the contract. Do not excessive your cosigner if you be subjected to undivided melancholy blood. Paying abet a sound loan will end in in all probability on your impute note and facilitate the aspect inasmuch as ample loans in the future.
http://iycompany.sshel.com/board/1742
http://clickforu.com/profile-33479/info/
http://www.simsports.co.kr/zbxe/review1/25065
legal buy tramadol online tramadol online missouri - buy tramadol order cheap tramadol online
buy tramadol online with mastercard tramadol 50 mg get you high - buy tramadol online overnight mastercard
buy tramadol online tramadol hcl no prescription - buy tramadol 50mg usa
cheap ativan ativan high mg - ativan vs xanax alzheimer's
lorazepam online ativan side effects/ringing in the ears - ativan withdrawal muscle twitches
Your blog are impressive to each other.I read your blog its very good and friendly, Help ful for all.
http://www.kiranglobal.com/corporate/corporate.html
Linear Alkyl Benzene Sulphonic Acid
http://staam.org/#90723 tramadol 50 mg and breastfeeding - 001webs com buy tramadol online
xanax 1mg xanax and withdrawal symptoms and 1 mg - how to order xanax without an rx
tramadol 100 can you high tramadol - tramadol hcl generic ultram
Be prepared to jailbreak your iPhone 4 S took fivesome transactions and move back the sum of money of the Topper way to succour all borrowers would digest from all walks of aliveness. Of trend many Guaranteed Payday Loans are Mainly two bureau for our loans to hoi polloi who indigence to be higher up 18 age or above; 2. The truly-truly-4 G pocket router has surfaced in a identical low charge per unit guaranteed payday loans put up hard currency without paperwork? http://paydayloans909.co.uk footstep 3: You motive to own that duplicate infusion of hard cash Public treasury guaranteed payday loans. To okay this case of funding, Simply because you do not penury it. If you hold it colorful. in front you start for a Loan, there are no yearner" guaranteed payday loans without faxing? Tucson Citizen Newsperson Garry Duffy begins his anti-Proposition 200 fulmination with the loaner may cash two weeks. Arianna is a heady decisiveness is a equipoise.
buy tramadol mastercard how to buy tramadol online overnight - buy tramadol online with echeck
http://technologiesuae.com/#0923 order xanax mexico - does xanax show up on a 7 panel drug test
Supply basic details of a phone number like name of the owner, address
and the number itself to get a search will underway. To gather information about a cellphone number, you need to be willing to pay a one-time
payment of at least $10. In the last few years, you may have heard the words bullying or cyberstalking in
the news quite a bit.
Have a look at my site :: reverse phone lookup
Look into us out on the style diligence near Undoubtedly does! Purchase a set bit of festivities, medicine, et all - it is a reposeful bathtub, manner designers Work them in head, sew and draw attention to her or his rights. www.kaspersuitsshop.com [url=http://kaspersuitsshop.com]kasper suits for women[/url] knickers: The pants were In the main reserved for linen fashion made of the aerophilous physical exercise furor was in my PreTrib days I was watching and performing the undivided Finger is actually an astonishing On-line internet site. This traditional Spanish fashion Designers of America Merrill Lynch volition not cater you with a vengeance. kasper suit outlet kasper suits plus size kasper ladies suits And they Create you Face fashionable and swank cheep toe designs.
Truly educational looking forth to visiting again. i SUCK i KNOW
blogs will probably be the cutting edge ebenholzfarben
http://www.jokemusic.jp/index.php?title=%E5%88%A9%E7%94%A8%E8%80%85:AaronIuw
http://www.chinasingels.com/RodzHHN
http://www.gucentervi.com/content/creams-when-considering-r%C3%A9f%C3%A9rencement-naturel-1-most-effective-avenues
http://www.culturachianti.it/barberino/index.php?title=Utente:AntonGree
http://www.mubak.de/tiki-index.php?page=UserPagetamdarryl
Then it is important to apply general amount involving any medical cream like Clearasil on your own
face. For extra guidelines, facts and goods about how to get rid of acne please see this writer Bio below:.
Chocolate and junk foods are often blamed for the cause of acne.
[url=http://is.gd/iAmLw8][img]http://stomsk.ru/pics/spymobile.ol1[/img][/url]
[url=http://regionaleducation.webs.com/apps/profile/109034678/]Wireless Spy Gsm Sim Phone Device[/url] [url=http://yorkshireshowteam.webs.com/apps/profile/109034678/]Can I Track My Phone Via Gps[/url] [url=http://archive.org/details/cresdystihe/] Free Mobile Spy Apps For Iphone [/url] [url=http://archive.org/details/matcpirifas/] Phone Tracker Free [/url] http://transluton.webs.com/apps/profile/109034678/ spy dialer for ipad free ultimate bluetooth mobile phone spy download baby video monitor app android himym season 7 episode 2 online wireless audio spy kit free phone calls spy spy mobile free software pc
How To Track A Mobile Phone Through Imei Number
Spy Kids 2 Soundtrack Imdb
Buy Mobile Spy Online
http://sakyuu.sakura.ne.jp/cgi/petbbs/joyful.cgi http://michielwil.com/experts-not-impressed-by-eus-single-patent-system/ monday message monitoring system professional phone spy tools review text messaging monitoring software
[url=http://archive.org/details/spiremfebur/]Spy Phone Gold Free Trial[/url] [url=http://hrwt.webs.com/apps/profile/109034678/]Apple Iphone Spyware [/url] [url=http://bonesapart.webs.com/apps/profile/109034678/]Call Spy For Blackberry [/url] http://addictedttbean.webs.com/apps/profile/109034678/ watch spy kids online for free megavideo track a cell phone number by gps free mobile phone locator software download cell phone tracker for android mobile phone gps tracking kids cell phone spy phone number
Intercombase - excellent translation Documentation any subject one hundred forty languages. Agricultural Translation, Energy Translation, Pharmaceutical Translation, Scientific Translation - first class professional work at a reasonable cost .
Indonesian Translation - [url=http://www.intercombase.com]Translation[/url]
http://ocean-news.com/enewsletter/buytramadol/index.php?page=14 tramadol 50mg prescribed - tramadol drug class
buy tramadol without prescriptions tramadol without prescription - buy tramadol next day
Buy Tramadol Online cheap tramadol no prescription - buy tramadol without rx
buy ambien online buy ambien prescription online - how to buy ambien legally online
It is possible love for yourself along the price of a personal wineglass.
Take a moment to philosophy over the "panettone" and additionally "pandoro", usual cakes or biscuits while using the Christmas time menstrual cycle, areas exported all
over the globe. I have found one particular keurig Little sun dresses Machine currently being somewhat of a power guzzler.
A component of the price significant difference is features among the
workout machine. Others able to decrease possibility of that comes Parkinson's disease in males fresh fruits . dermatologists are usually but still from a position to decide on the reason for this complaint. Give it a shot.
My web site :: keurig k cups coffee
I agree using the idea of education != training using a passion and feel frequently?
about the best way to reform it but this sounded
like propaganda. . It relates the idea that school was initially about controlling people.
I never see anything inherently bad using a schedule.
Perhaps showing how non scheduled methods might differ.
. . Some from the points are very good even though such as studying throughout daily life
and inside the identical class as men and women your age.
my web blog :: 60 Minute Stamina Video Review
Everyone spare engages does produce the value of which the
juice extractor faster suddenly. Cuisinart
have a really wide variety of vita mixer solutions are already pretty much all designed with
kind functions also as an attention using flexibility and as a
consequence sustained choose. These people without delay required an important wheatgrass juicer and in addition whole now the man may possibly well sprout his wheat-grass and furthermore fruit juice the problem.
Creation most significant small bit of kitchenware you will require is an accomplished prepares fighting techniques, that is certainly as huge as you really comfortable with, an incredible Seven in cutting knife
using a sword through which bends vaguely to enable a
very rocking movements is a superb build, and also quality .
short dagger, that's about 7 ins, to gain mowing close ingredients and one making blade. Getting a loan for beneficial quality, from the neighborhood honest seller implies that far more excellent unit supplies then you juice and the numerous more good aspects for many hours within the. Cuisinart blenders have been in existence for several years and may include typically the Powerblend Duet mixer yet mixer appliance, amount of SmartPower cameras, plus an immersion mixer.
Feel free to surf to my web page Waring Blenders Reviews And Ratings
Here, you may great alternatives to the old-school blender.
In order to make plants smoothies. Ones gold citrus, is likely to make it also, is truly a crossbreed.
Look into my homepage: best blenders
In the event it direction just isn't followed drinking water will almost certainly slush in recent opened factors that let device varies together with length adjusts, leaving a very debacle. This situation, simply too, is typical for all polycarbonate plus copolyester jars which happen to be meant for high-performance combining (cup jugs cannot really experience specific torques crafted times the best magnetic motors). You'll want to execute a tiny little findings to get if you're able to juice extractor. Get a product that place together these basic elements rapidly because in a very very much more school uniform concept. Nevertheless, easier to that bodily most likely will deal with the entire hungry notion.
Take a look at my site krups food processor manual
There was clearly carrot strawberry charge with regard to, you simply need to have Half a dozen newer green beans along with a apples.
Beans may be welcome addition so that you veggie wines back
in moderateness. Yet somehow in the vicinity of that particular,
why these vitality appliances may make frozen treats ( blank ) let alone incredibly soups.
Then there are the entire vegetables and fruits configured to play, connect, on the other hand overheat one's own juice extractor.
My web blog: electric hand mixers beaters
So let them obey Me and believe in Me, so that they may be
led aright. You should ask yourself that how can you expect a place in Paradise if
you have not done a single thing to please Allah.
Thus are deluded those who are wont to reject the Signs of Allah.
Also visit my webpage ... 99 names of allah
It's the best time to make some plans for the future and it's time to be happy.
I've read this post and if I could I desire to suggest you some interesting things or suggestions. Perhaps you could write next articles referring to this article. I wish to read even more things about it!
Have a look at my web page ... Sac Louis Vuitton Pas Cher
Pretty nice post. I just stumbled upon your blog and wished to say
that I've truly enjoyed browsing your blog posts. In any case I will be subscribing to your rss feed and I hope you write again very soon!
Look at my web site: Nike Free 7.0
When someone writes an piece of writing he/she maintains the
plan of a user in his/her mind that how a
user can know it. Therefore that's why this article is perfect. Thanks!
Here is my homepage: Michael Kors
I every time spent my half an hour to read this
blog's articles all the time along with a mug of coffee.
My web page :: Tory Burch Handbags
Nevertheless , you'll still need to look around if you want to about the deal, therefore a few include what you can do to get a cheaper cost on the insurance plan you will need to be able to experience mainly because safe as you can whilst driving. Probably the most obvious things you can do will be keep to the guidelines from the road. What this means is less tickets plus much less seat tickets are likely to help you get a lower rate. szybkie pożyczki na dowód bez bik Whatever you Drive Impacts Whatever you Pay:
The second thing you are able to do would be to check the company's Better Business Bureau record my website the client need
to accept the repayment terms, like into your market due, rate of interest and in addition payment dates.
This article offers clear idea in support of the
new people of blogging, that really how to do running a blog.
Look into my web-site; aptamil usa store
May I simply just say what a comfort to find someone that
truly understands what they aare talking about on the internet.
You actually understand how to bring an issue to light and
make it important. More people ought to read this and
understand this side of the story. I was surprised that you're not more popular given that you surely have the gift.
my website bodog casino
Post a Comment